PROTEINDEX™ Glutathione Agarose 4 FF, Prepacked Cartridge
Catalog NO.
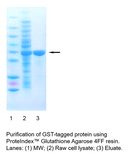
Convenient, efficient, one-step chromatography purification of GST-tagged fusion proteins with high yield and purity. Compatible with syringe, AKTA, and peristaltic pump systems. ... Read more
Description
PROTEINDEX™ Glutathione Agarose 4FF is an affinity chromatography medium developed for easy, one-step purification of recombinant glutathione S-transferase (GST) fusion proteins and other glutathione binding proteins expressed in E. coli, insect and mammalian cells. The rigid, highly cross-linked support is suitable for purification scale-up applications. The cartridge prepacked with Glutathione Agarose 4FF is ready-to-use, and is compatible with syringe, AKTA, and other peristaltic pump systems.
Value bundle with a Luer Female/10-32 Male Connector in PEEK (Cat# 20-0315) is now available for all pack sizes, providing convenient operation of the pre-packed column with a syringe at an excellent value.
Product Details
| Ligand | Glutathione coupled with a 12-atom linker |
| Ligand concentration |
120 - 320 μmol Glutathione/mL medium |
| Binding capacity | >10 mg GST (26 kDa)/mL medium |
| Matrix | 4% highly cross-linked agarose |
| Particle size | 45 μm - 165 μm |
| Maximum pressure |
0.3 MPa (3 bar, 43 psi) |
| Recommended flow rate |
300 - 500 cm/hour |
| pH stability |
3-12 |
| Storage temperature |
2 - 8°C |
| Storage buffer |
20% ethanol as preservative |
Contact Us
Marvelgent Biosciences Inc.
Head Office
303 Wyman St, Waltham, MA 02451, USA
Telephone
1.888.330.6623
Fax
1.888.330.6623
Emails
Ordering support: order.support@marvelgent.com
Technical support: cust.support@marvelgent.com
Careers: career@marvelgent.com
You are welcome to leave a message using our Contact Form. We will get back to you within 24 hours.
We Also Recommend