PROTEINDEX™ Ni-Penta™ Agarose, Chemical Stable
Catalog NO.
Single-step IMAC purification of polyHistidine-tagged proteins in the most suited conditions. EDTA resistant, β-ME compatible affinity for greater purity. ... Read more
Description
PROTEINDEX™ Chemical-Tolerant Ni-Penta™ Agarose is a new immobilized metal ion affinity chromatography (IMAC) medium precharged with nickel ions, designed mainly for purification of low abundant proteins or challenging proteins in conditions where EDTA, DTT, β-ME, and other stabilizing agents are necessary for maintaining protein stability and integrity.
The nickel ion binds very strongly to the matrix support in complex with a proprietary pentadentate chelating ligand, which immobilizes the nickel ion through 5 sites to form a homogeneous, chemical-stable structure. This characteristic nickel ion bonding on the Ni-Penta™ resin provides excellent resistance to EDTA, NaOH, β-ME, and other reagents that are restricted in the purification protocol using conventional NTA- and IDA- functionalized resins.
Product Highlights
- EDTA-resistant
- High selectivity for high purity
- Retained high 6xHis affinity in various buffers
- Well suited for direct capture of secreted polyHis-tagged proteins from eukaryotic cell culture supernatant
- Recharge-free with minimal nickel ion leakage
- Requires less imidazole
- NaOH-tolerant
- Versatile, ready-to-use with convenient formats
Applications:
- Purification of secreted His-tagged proteins directly from eukaryotic cell culture supernatants
- Owing to its proprietary ligand for nickel ion immobilization, Ni-Penta™ resin tolerates EDTA very well, and its affinity for His-tag can be retained in presence of EDTA up to 10 mM. To purify secreted proteins that are produced from eukaryotic cell cultures (such as insect High Five™ cells* and Sf9 cells, CHO cells, 293 cells etc.), a step of removing incompatible agents to Ni-NTA resins by dialysis is frequently required prior to loading the material on the column. This is tedious, time-consuming, and labor intensive. By using Ni-Penta resins, sample pretreament can be skipped, and the culture medium can be loaded directly to the column for purification.
- Purification of His-tagged proteins that require presence of EDTA, DTT, β-ME, and other agents for maximum protein stability and integrity
- PROTEINDEX™ Ni-Penta™ Agarose Resin is compatible with a wide range of buffers including chelating agents (e.g. EDTA) and reducing agents (e.g. DTT), which are incompatible and therefore should be avoid in purification applications using conventional NTA or IDA resins. Ni-Penta™ resin helps to simplify your sample preparation process and allows your target proteins be purified at their preferred conditions.
- Repetitive uses of the column that require clean-in-place with NaOH but no nickel recharge
- Anti-strip power of the Ni-Penta™ enables repetitive uses of the column without the need to recharge nickel ion, even after multiple cycles of CIP with 1 M NaOH.
- Restricted imidazole amount in the elution buffer due to the requirements in downstream applications or the target protein's sensitivity to imidazole
- Imidazole is general required to minimize non-specific binding on Ni-NTA resins. Ni-Penta™ Agarose Resin is highly selective for His-tag and therefore does not demand imidazole for minimizing non-specific binding throughout purification. No imidazole is required in the binding buffer, and 40 mM to 100 mM of imidazole can efficiently elute most target proteins.
Technical Notes
1. EDTA-stable Ni-Penta chemistry for minimal nickel ion leakage

Figure 1. Strong chelate chemistry for immobilization of nickel ion. Ni-Penta Agarose 6 FF resins were incubated at room temperature for 2 hours in 5 volumes of a serial dilutions of EDTA from 10 mM to 400 mM. The amount of nickel ions released into the buffer was measured.
Result: During 2 hours of incubation in 50 mM EDTA, less than 5 % of nickel ions leaked from Ni-Penta resins. In comparison, 50 mM EDTA is sufficient to strip off nickel ions from conventional Ni-NTA resins and has been used routinely to regenerate Ni-NTA resin.
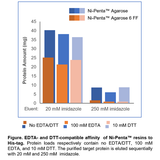
2. Uncompromised His-tag protein binding in EDTA up to 10 mM

Figure 2. Retained His-tagged protein binding capacity in presence of EDTA. Ni-Penta 6FF resins were added to 20 volumes of E.coli lysate (~0.5 mg/mL containing a His-tagged protein of 26 kDa in size), in the presence of 2 mM, 5 mM, 10 mM EDTA respectively. Protein binding was then performed at room temperature for 2 hours. After washing 5 times with 20mM sodium phosphate, 0.5 M NaCl, 5 mM imidazole, pH7.4, the target His-tagged protein was eluted with 20mM sodium phosphate, 0.5 M NaCl, 250 mM imidazole, pH7.4. Protein concentration was measured by UV absorbance at OD280.
Result: In the presence of 10 mM EDTA, the amount of His-tag protein being purified from Ni-Penta resin was similar to those being purified in the presence of less or no EDTA.
3. Tolerance to reducing agents

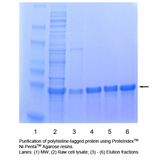
Figure 3. Tolerance to reducing agents. Ni-Penta 6FF resins were added to 20 volumes of E.coli lysate (~0.5 mg/mL containing a His-tagged protein of 26 kDa in size), in the presence of various dilutions of reducing agents, including TCEP, DTT, and β-ME respectively. Protein binding was then performed at room temperature for 2 hours. After washing 5 times with 20mM sodium phosphate, 0.5 M NaCl, 5 mM imidazole, pH7.4, the target His-tagged protein was eluted with 20mM sodium phosphate, 0.5 M NaCl, 250 mM imidazole, pH7.4. Protein concentration was measured by UV absorbance at OD280.
Result: In the presence of up to 5 mM TCEP, 10 mM DTT, or 50 mM β-ME, the amount of His-tag protein being purified from Ni-Penta resin was similar to those being purified in absence of the respective reducing agents.
(A) The percentage of retained protein binding capacity is plotted against the concentration of individual reducing agents. (B) Representative SDS-PAGE analysis of the eluates.
Note:
* High Five™ is a trade mark of Thermo Fisher Scientific.
Product Details
| Ligand | Nickel |
| Binding capacity | >10 mg 6XHis-tagged protein/mL medium |
| Matrix | 4% agarose supplied as 50% slurry |
| Particle size | 45 μm - 165 μm |
| Maximum pressure |
0.1 MPa (1 bar) |
| Recommended flow rate |
Gravity flow, <75 cm/hour |
| Chemical compatibility |
|
| Storage temperature |
2 - 8°C |
| Storage buffer |
1X PBS containing 20% ethanol |
Product is shipped at ambient temperature. Upon receipt store at 2 - 8°C. Do not Freeze.
![]()
![]()
Documents
Product Information Sheet
Accessories
Disposable Gravity Flow Columns
Contact Us
Marvelgent Biosciences Inc.
Head Office
303 Wyman St, Waltham, MA 02451, USA
Telephone
1.888.330.6623
Fax
1.888.330.6623
Emails
Ordering support: order.support@marvelgent.com
Technical support: cust.support@marvelgent.com
Careers: career@marvelgent.com
You are welcome to leave a message using our Contact Form. We will get back to you within 24 hours.
We Also Recommend