ProteoSure™ Recombinant Bovine Enterokinase
Catalog NO.
Ultra pure, animal orgin-free EK endopeptidase specifically cuts at sequence DDDDK with high activity. ≥5 U/μL. Long-lastingly cleaves at low temp 2 - 8 °C. ... Read more
Description
Enterokinase is a specific protease that cleaves at the carboxyl side of Lysine residue when it is preceded by four Aspartic acids (Asp-Asp-Asp-Asp-Lys) and is not followed by Proline. Enterokinase can be used to remove the fusion tag of a protein or the N-terminal pro-sequence of a precursor protein that is genetically engineered to be activable with enterokinase.
ProteoSure™ Recombinant Bovine Enterokinase is produced from E.coli. and contains no trace of contaminating proteases. The enzyme is purified by a proprietary process to obtain high purity and maximum enzymatic activity. The product is delivered as clear aqueous solution (≥ 5 U/μL) in a storage buffer specifically formulated to ensure the highest enzyme stability during storage.
The enzyme is highly active at 4°C, allowing digestion to be performed at a desirable low temperature for maximum stability of temperature-sensitive target substrates during digestion.
Unit definition: One Unit is defined as the amount of enzyme required to cleave 0.5 mg of a DDDDK-containing protein (~30 kDa in size) to 95% completion in 12 to 16 hours at 25°C.
Product Highlights
- High activity
One unit is able to cleave 0.5 mg of a DDDDK-containing protein to 95% completion in 12 to 16 hours at 25°C. It is supplied as concentrated solution of > 5U/µL.
- High purity
There are no any other protease contaminants nor animal-derived impurities.
- High specificity
- Protease that cleaves specifically after a lysine preceded by four aspartic acids: Asp-Asp-Asp-Asp-Lys (DDDDK)
- No detectable off-target cleavage when used at 2 - 25°C.
- Long-lasting superior performance. Retained enzymatic activity at 2 - 8°C.
It allows low-temperature digestion to ensure stability of temperature-sensitive target proteins
- Compatible with the following reagents:
Imidazole (<100 mM), NaCl (<50 mM), and glycerin (<5%).
Applications
- Fusion tag removal from the recombinant peptide fragment or protein that is preceded by the characteristic sequence DDDDK
- Cleavage or processing of engineered proteins that contain enzyme recognition sequence.
- Processing enzymes and proteins that are unstable in room temperature
Technical Notes
Recommendations for fusion protein digestion
Reaction buffer: 25mM Tris-HCl, pH8.0
Fusion protein concentration: 0.1 ~ 1 mg/ml (total protein content: 0.5-1.0 mg)
EK content: 1-2 U per reaction
Temperature: 25°C
Incubation time: 12h-16h or overnight
Chemical stability
Certain chemicals and reagents may affect Enterokinase activity, including:
Imidazole (>200 mM), NaCl (>200 mM), and glycerin (>5%)
It is necessary to decrease the concentration of agents that may interfere enterokinase activity prior to enzyme digestion. Proceed with one of the followings:
- To achieve optimum result, dialyze the sample against 25 mM Tris-HCl (pH 8.0) before digestion;
- If dialysis is undesirable, dilute the sample to a final solution that contains <100 mM imidazole, <50 mM NaCl, and <5% glycerin. Maintain the ratio of EK to fusion protein at 1 U for every 0.5 mg fusion protein in the reaction;
- If the sample solution contains one or more components which concentrations are higher but further dilution or depletion is not desirable, it is also recommended to increase the EK amount in the reaction and to extend the reaction time.
Optimum reaction temperature for enzymatic activity
It is recommended to perform enzyme digestion at a temperature between 4 - 25°C, as the enzyme remains active for extended period without showing apparent non-specific cleaving activities.
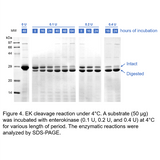
Showed in Figure 1, recombinant bovine enterokinase was used to cleave a substrate at 4°C. The enzyme cleaves at the specific site, and yields a fragment of the expected size. 0.4 Unit of EK is able to digest to 90% completion after 24 hours at 4°C.
 |
Figure 1. EK cleavage reaction under 4°C. A substrate (50 µg) was incubated with enterokinase (0.1 U, 0.2 U, and 0.4 U) at 4°C for various length of period. The enzymatic reactions were analyzed by SDS-PAGE.
Recombinant bovine enterokinase shows optimum cleavage activity at 25°C, without detectable nonspecific enzymatic activities (Figure 2). When incubated for 24 hours at 25°C, 0.1 U of EK is sufficient for complete digestion of 50 µg substrate. When enzymatic reaction was extended to 64 hours, only one protein fragment of the expected product size was revealed, suggesting no cleavage outside of the specific enzyme-recognition sequence.
 |
Figure 2. Optimum EK cleavage reaction occurs at 25°C. A substrate (50 µg) was incubated with 0.1 U enterokinase at 25°C for extended period of time, from 8 hours to 64 hours. The resulted protein fragmants after enzymatic reactions were analyzed by SDS-PAGE. Incubation of the substrate with 0.1 U of EK for longer period resulted digestion to completion, without nonspecific cleavage. No degradation of the digested substrate was detected.
When the enzyme reaction is performed under an elevated temperature, such as 37°C, the substrate was shown to be degraded after 8 hours of incubation, suggesting that nonspecific cleavage occurs outside of the specific EK recognition site (Figure 3).
 |
Figure 3. EK is active at 37°C but shows certain degrees of non-specific enzymatic activities after extended period of incubation. A substrate (50 µg) was incubated with 0.1 U enterokinase at 37°C for 2 hours to 24 hours. After 8 hours of enzyme digestion at 37°C, the substrate disappeared on SDS-PAGE as a result of degradation.
Product Details
| Synonyms | Enteropeptidase |
| CAS # | 9014-74-8 |
| Enzyme Commission # | 3.4.21.9 |
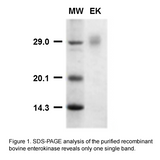
| Molecular weight | Theoretical MW: 25.8 kDa Apparent MW on SDS-PAGE: ~ 27 kDa |
| Origin of species | Bovine |
| Source | E.coli |
| Appearance | Clear aqueous solution, filter-sterilized |
| Specific activity |
≥5 U/μL |
| Unit definition | One unit is defined as the amount of enzyme needed to cleave 0.5 mg of a special fusion protein in 12 to 16 hours to get 95% completion at 25°C in a buffer containing 25 mM Tris-HCl (pH 8.0). |
| Storage & stability | The product is shipped with blue ice. Store at -20°C upon receipt. It remains stable without detectable loss of activity after one week at 25°C. Stable after several freeze-thaw cycles. |
| Intended use | For research or further processing only. Not intended for any human or animal diagnostic or therapeutic use. |
Documents
Product Information Sheet
🗎 Recombinant Bovine Enterokinase
Technical Note
Contact Us
Marvelgent Biosciences Inc.
Head Office
303 Wyman St, Waltham, MA 02451, USA
Telephone
1.888.330.6623
Fax
1.888.330.6623
Emails
Ordering support: order.support@marvelgent.com
Technical support: cust.support@marvelgent.com
Careers: career@marvelgent.com
You are welcome to leave a message using our Contact Form. We will get back to you within 24 hours.
We Also Recommend